If you are searching about Why transparency is required in the reporting of medical device failures you've visit to the right web. We have 7 Images about Why transparency is required in the reporting of medical device failures like Why transparency is required in the reporting of medical device failures; Jabil Circuit on the Forbes Global 2000 List and also Our Company â€" Siltronic / perfect silicon solutions. Here it is:
Why Transparency Is Required In The Reporting Of Medical Device Failures
 www.nsmedicaldevices.com
www.nsmedicaldevices.com failures
Http://www.gogofinder.com.tw/books/pida/6/ OPTOLINK 2013 Q2光連國際版å£åˆŠ
Canada Ranks Above Global Average For Industrial Robot Density: IFR
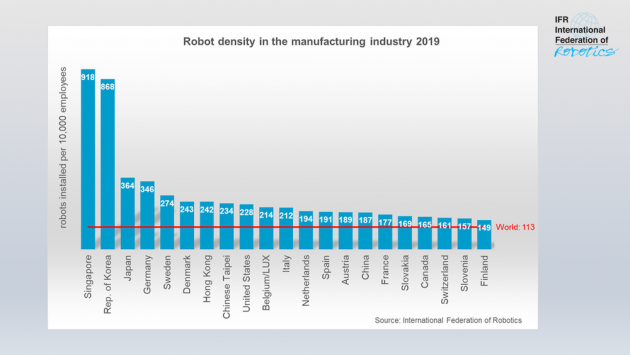 www.automationmag.com
www.automationmag.com density ifr automated industrial roboter robotisation ranks nationer blandt verdens stadig producer azspot glgt industrierobotik wichtigsten liegt glade releases angulaire
 venturebeat.com
venturebeat.com SK Innovation's First Lithium-Ion Cell Plant In China Is Complete
 insideevs.com
insideevs.com plant batterie lithium bannata gigafactory dagli delle
Jabil Circuit On The Forbes Global 2000 List
 www.forbes.com
www.forbes.com jabil
Our Company â€" Siltronic / Perfect Silicon Solutions
Density ifr automated industrial roboter robotisation ranks nationer blandt verdens stadig producer azspot glgt industrierobotik wichtigsten liegt glade releases angulaire. Http://www.gogofinder.com.tw/books/pida/6/ optolink 2013 q2光連國際版å£åˆŠ. Why transparency is required in the reporting of medical device failures


